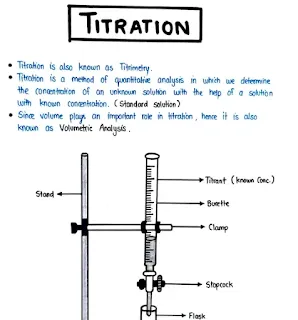
Unit-2 Pharmaceutical Analysis Handwritten Notes: Acid-Base and Non-Aqueous Titrations
Download these handwritten notes covering Unit-2 of Pharmaceutical Analysis, focusing on Acid-Base and Non-Aqueous Titrations. These notes provide a simplified overview of the theories, classification, and applications of these titrimetric methods.
Keywords: Pharmaceutical Analysis, Unit-2, Handwritten Notes, Acid-Base Titration, Acid-Base Indicators, Neutralization Curves, Non-Aqueous Titration, Solvents, Acidimetry, Alkalimetry, Sodium Benzoate, Ephedrine HCl, PDF Download, Study Material.
Understanding Titrimetric Analysis: Unit-2 Pharmaceutical Analysis Handwritten Notes on Acid-Base and Non-Aqueous Titrations
Unit-2 of Pharmaceutical Analysis delves into the principles and applications of titrimetric analysis, specifically focusing on Acid-Base and Non-Aqueous Titrations. These handwritten notes offer a concise and accessible guide to understanding the theories, classifications, and practical applications of these essential analytical techniques.
Acid-Base Titrations: Quantifying Acids and Bases
Acid-base titrations are a fundamental quantitative technique used to determine the concentration of an acid or a base. The notes cover the following important aspects:
- Theories of Acid-Base Indicators: Explanation of how acid-base indicators work, including the role of pH and color change.
- Classification of Acid-Base Titrations: Categorization of different types of acid-base titrations, such as strong acid-strong base, weak acid-strong base, strong acid-weak base, and weak acid-weak base titrations.
- Theory Involved in Titrations of Strong, Weak, and Very Weak Acids and Bases: Detailed discussion of the equilibrium principles and calculations involved in each type of titration.
- Neutralization Curves: Understanding and interpreting neutralization curves, which show the pH change during a titration and are used to determine the endpoint.
Non-Aqueous Titrations: Titrations in Non-Aqueous Solvents
Non-aqueous titrations are used when the analyte is insoluble in water or when the reaction does not proceed satisfactorily in aqueous solution. The notes cover:
- Solvents Used in Non-Aqueous Titration: Discussion of different types of solvents used in non-aqueous titrations, such as protic, aprotic, and amphiprotic solvents, and their properties.
- Acidimetry and Alkalimetry in Non-Aqueous Solvents: Explanation of how acidimetry (titration of bases) and alkalimetry (titration of acids) are performed in non-aqueous solvents.
- Estimation of Sodium Benzoate: Application of non-aqueous titration for determining the concentration of Sodium Benzoate.
- Estimation of Ephedrine HCl: Application of non-aqueous titration for determining the concentration of Ephedrine HCl.
Benefits of Handwritten Notes:
These handwritten notes provide a simplified and easily accessible resource for students and professionals studying pharmaceutical analysis. They offer a concise overview of the key concepts and techniques related to acid-base and non-aqueous titrations, making them a valuable tool for exam preparation and quick review.
Download these Unit-2 Pharmaceutical Analysis handwritten notes to enhance your understanding of acid-base and non-aqueous titrations and their applications in pharmaceutical analysis.
Info!
If you are the copyright owner of this document and want to report it, please visit the copyright infringement notice page to submit a report.